|
11/7/2019 Woodward Fieser Rules Pdf
Calculation of λ using woodward fieser rules. 1.
Slide 5: 5 The Woodward-Fieser rules are designed to predict the absorbance of conjugated systems in the ultraviolet-visible region of the spectrum based on the connectivity of atoms within the system. It is used for finding absorption maxima position (λMax). This may be differs from observed values by 5-6. Woodward fieser rules pdf Woodward fieser rules pdf Fieser-Kuhn Rules Woodward-Fieser rules for dienes work well for conjugated systems with less than 4 double bonds in. Check out this post for to learn how to use these rules.
Assignment #2 Calculation of λmax using Woodward-Fieser rules. By Sayyad Ali To Prof. Jamshed Iqbal CIIT Abbottabad. Introduction to Woodward-Fieser Rules In the middle of the last century, R. Woodward studied the UV spectra of conjugated dienes and developed a set of rules for predicting the wavelength of maximum UV absorption based on the structure of the diene. Fieser extended the rules to conjugated aldehydes and ketones. In order to apply the rules to specific structures, we need to learn how certain structures are described, so we may apply numeric wavelength values to the structure.
Conjugated Dienes We know that conjugated dienes must lie in an s-cis conformation in order to undergo a Diels- Alder reaction. Thus, two fundamental conformations of conjugated dienes are the s-cis conformation and the s-trans conformation. Figure 1 shows that an s-trans conjugated diene is one in which the two double bonds lie on opposite sides of the single bond that joins them. Whereas, a double bond in the s-cis conformation is one in which both double bonds lie on the same side of the single bond that joins them. 1,3-butadiene s =single bond that connects the two double bonds (dashed, straight line below) s-trans conformation (double bonds on opposite sides of single bond) s-cis conformation (double bonds on same side of single bond) Figure 1.
Conformations of 1,3-butadiene. The first-step in predicting the wavelength of maximum UV absorption for a conjugated diene is to determine whether it lies in an s-trans or s-cis conformation. If it lies in the s-trans. conformation, its base wavelength is 217 nm. If it lies in the s-cis conformation, its base wavelength is 253 nm. Endocyclic vs Exocyclic Double Bonds An interesting feature of double bonds is that they may be part of a ring system, in which case they are called endocyclic double bonds because their bond lies “within” the ring.
Double bonds may also project from a ring, in which case they are called exocyclic double bonds because their bond lies “outside” the ring. If a compound is bicyclic, a double bond might be endocylic with respect to one ring and exocyclic with respect to the other ring.
Endocyclic exocyclic A B endo to A exo to B Figure 2. Types of double bonds. Figure 2 shows that a double bond is endocyclic if its bond is part of the ring. The double bond is exocyclic if its bond projects from the ring. If a double bond is exocyclic to a ring, it adds 5 nm to the base wavelength of a conjugated diene. Extended Conjugated Double Bonds Two double bonds separated by a single bond are conjugated.
If a third double bond is separated from one of the original pair of double bonds by a single bond, the three double bonds represent an extended conjugated system. s-cis max) = 217 nm s-cis extended max) = 247 nm s-trans max) = 253 nm s-trans extended max) = 283 nm Figure 3. Effect on λmax of extending the conjugation. Each double bond that extends the conjugation adds 30 nm to the wavelength of maximum absorption.
Effect of Alkyl Groups Any alkyl group bonded to a carbon atom of the conjugated system (i.e., a carbon sharing a conjugated bond) adds 5 nm to the wavelength of maximum absorption. S-cis + 2 alkyl groups max) = 227 nm s-cis extended + 3 alkyl groups max) = 262 nm s-trans + 2 alkyl groups max) = 263 nm s-trans extended + 3 alkyl groups max) = 298 nm Figure 4. Conjugated systems containing alkyl groups. The wavelength values shown in figures 3 and 4 are predicted values. The actual values vary slightly from the predicted values and must be determined by experimentation.
Conjugated Aldehydes and Ketones A conjugated aldehyde or ketone arises when a double bond is separated by a single bond from the carbonyl group of an aldehyde or ketone. O H O conjugated aldehye (max) = 210 conjugated ketone (max) = 215 Figure 5. Conjugated aldehyde and ketone.
The base wavelength for a conjugated aldehyde is 210 nm and for a conjugated ketone is 215 nm. The compounds shown in Figure 5 are also called -unsaturated because their carbon- carbon double bond lies between the alpha and beta carbon atoms. The Greek lettering system starts with the carbon atom bonded to the carbonyl carbon atom of the aldehyde or ketone. An alkyl groups bonded to an -carbon adds 10 nm to λmax and an alkyl group bonded to a - carbon adds 12 nm to the base value. The effects of an exocyclic double bond and extended conjugation are the same for aldehydes and ketones as for dienes. An exocyclic double bond adds 5 nm and an extended double bond adds 30 nm to the base values.
A wavelength predictor has been created in Excel for conjugated dienes and for conjugated aldehydes and ketones. A diene is characterized as lying in an s-cis or s-trans conformation and by its number of alkyl groups bonded to the conjugated system, exo double bonds, and extended double bonds.
The analyst enters these values into the predictor and the predictor calculates λmax. The predicted value of λmax is the same as one obtains by calculating the value as described above. A separate sheet in the Excel workbook handles aldehydes and ketones in a similar fashion. The structure is identified as an aldehyde or ketone, and its number of α and β alkyl groups, exo double bonds and extended double bonds are entered into the appropriate boxes. The predictor calculates λmax. Use the wavelength predictor to answer the problems in the two exercises found in the folder with this document.
The λmax of the p p. transition for compounds with.
Woodward Fieser Rules for UV spectrometry. 1. Assignment on ‘Woodward Fieser Rules’ Advanced Pharmaceutical Analysis (PHR 410) Submitted by, Md. Azamu Shahiullah Department of Pharmacy BRAC University. Assignment on ‘Woodward Fieser Rules’ Advanced Pharmaceutical Analysis (PHR 410) Submitted to, Ridwan Islam Department of Pharmacy BRAC University.
1 Department of Pharmacy BRAC University. Introduction 2. WOODWARD- FIESER RULES 3.1 Woodward Fieser rule for Conjugated Dienes and Polyenes 3.2 Parent values and increments for different Substituents or Groups 3.3 Example of Woodward Fieser rule for Conjugated Dienes and Polyenes 4.1 Woodward Fieser rule for α,β - unsaturated Carbonyl compounds. 4.2 Parent values and increments for different Substituents or Groups 4.3 Example of Woodward Fieser rule for α,β unsaturated hydrocarbon 5.1 Woodward Fieser rule for Aromatic compounds or Benzoyl derivatives. 5.2 Parent values and increments for Benzoyl Derivatives 5.3 Example of Woodward Fieser rule for Benzoyl derivatives. 1. Introduction In 1945 Robert Burns Woodward gave certain rules for correlating λ structure.
In 1959 Louis Frederick Fieser modified these rules with more experimental data, and the modified rule is known as Woodwar and λmax for a given structure by relating the position and degree of substitution of chromophore. WOODWARD- FIESER RULES Each type of diene or triene system is having a certain fixed value at which absorption takes place; this constitutes the Base value or Parent value alkyl substituents or ring residue, double bond extendi as –Cl, -Br etc are added to the basic value to obtain λ According to Woodward’s rules the λ λmax = Base value + Σ Su In 1945 Robert Burns Woodward gave certain rules for correlating λmax structure.
In 1959 Louis Frederick Fieser modified these rules with more experimental data, and the modified rule is known as Woodward-Fieser Rules. It is used to calculate the position for a given structure by relating the position and degree of substitution of FIESER RULES Each type of diene or triene system is having a certain fixed value at which absorption takes Base value or Parent value. The contribution made by various alkyl substituents or ring residue, double bond extending conjugation and polar groups such Br etc are added to the basic value to obtain λmax for a particular compound. According to Woodward’s rules the λmax of the molecule can be calculated using a formula: = Base value + Σ Substituent Contributions + Σ Other Contributions 3 max with molecular structure.
In 1959 Louis Frederick Fieser modified these rules with more experimental data, Fieser Rules. It is used to calculate the position for a given structure by relating the position and degree of substitution of Each type of diene or triene system is having a certain fixed value at which absorption takes. The contribution made by various ng conjugation and polar groups such for a particular compound. Of the molecule can be calculated using a formula: bstituent Contributions + Σ Other Contributions. 4 There are three sets of rules 1.
Woodward-Fieser rule for Conjugated dienes and polyenes. For - unsaturated Carbonyl compounds. For Aromatic compounds or Benzoyl derivatives. 3.1 Woodward Fieser rule for Conjugated Dienes and Polyenes a. Homoannular Diene:- Cyclic diene having conjugated double bonds in same ring. 1 2 3 4 5 6 b. Heteroannular Diene:- Cyclic diene having conjugated double bonds in different rings.
1 2 3 4 5 6 7 8 9 10. 5 c. Endocyclic double bond:- Double bond present in a ring. 1 2 3 4 5 6 d.
Exocyclic double bond: - Double bond in which one of the doubly bonded atoms is a part of a ring system. 1 2 3 4 5 6 7 8 9 10 A B Here Ring A has one exocyclic and endocyclic double bond. Ring B has only one endocyclic double bond. Double bond extending:- When more double bonds are present other than conjugations. 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 O 18 OH 19. 6 3.2 Parent values and increments for different Substituents or Groups Parent Value Acyclic conjugated dienes and Heteroannular conjugated dienes 215 nm Homoannular conjugated dienes 253 nm Acyclic trienes 245 nm Increments Each alkyl substitute or ring residue 5 nm Exocyclic double bond 5 nm Double bond extending conjugation 30 nm Auxochromes -OR 6 nm -SR 30 nm -Cl,-Br 5 nm -NR2 60 nm -OCOCH3 0 nm 3.3 Example of Woodward Fieser rule for Conjugated Dienes and Polyenes 1.
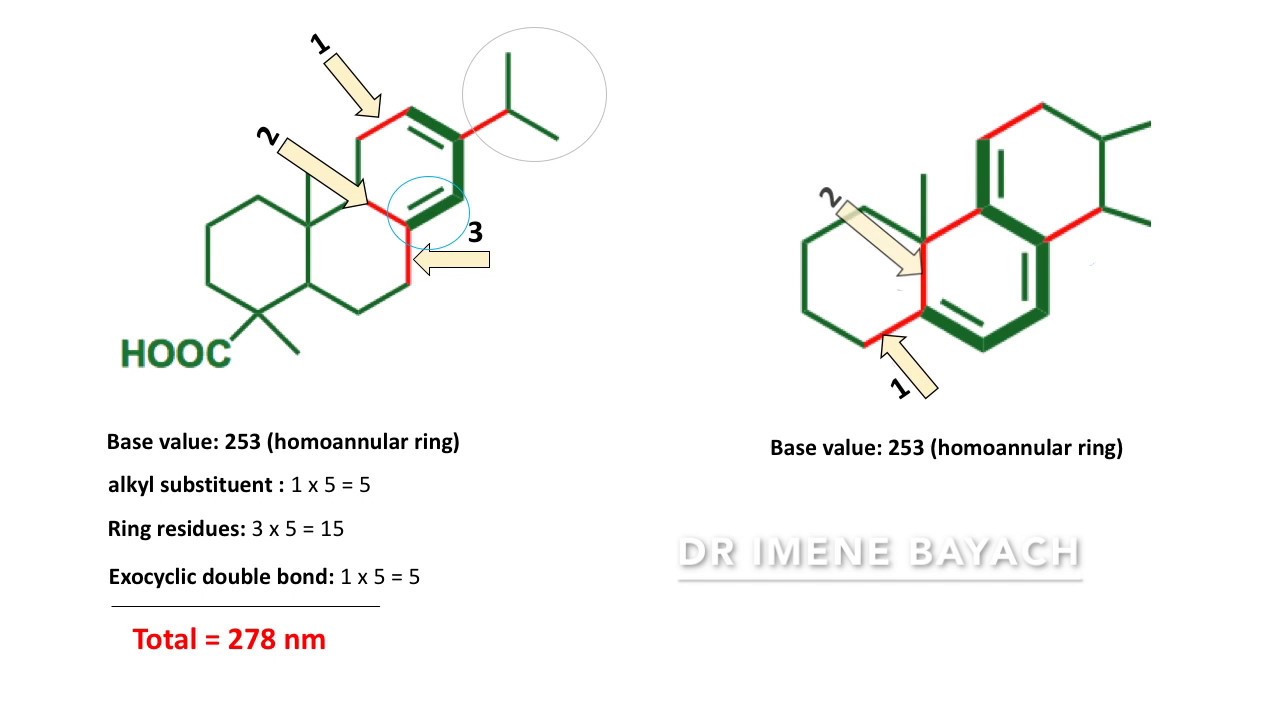
Example 1 (3E)-3,5-dimethylhexa-1,3-diene—ethane (1/2) Here, Parent value for Acyclic conjugated diene = 215 nm. 7 Alkyl Substitute or Ring residue at C-3 and C-5 = 5+5= 10 nm So, λmax would be = (215+10) nm = 225 nm Observed value = 225 + - 5 nm 2. Example 2 4a,7,8,9-tetramethyl-2,3,4,4a,4b,5,6,8a-octahydrophenanthrene Here, Parent value for Heteroannular conjugated diene = 215 nm. 8 Alkyl substitute or Ring residue = (5 x 4) nm = 20 nm Exocyclic double bond (in respect of B ring) = 5 nm So, λmax would be = (215+20+5) nm = 240 nm Observed value = 240 + - 5 nm 3. Example 3 1,2,4b,10-tetramethyl-1,2,3,4,4b,5,6,7-octahydrophenanthrene. 9 Here, Parent value Homoannular conjugated diene = 253 nm Alkyl substitute or Ring residue = (5 x 6) nm = 30 nm Exocyclic double bond (in respect with ring B) = 5 nm Double bond extending conjugation = 30 nm So, λmax would be = (253+30+30+5) nm = 318 nm Observed value would be = 318 + - 5 nm.
10 4.1 Woodward Fieser rule for α,β - unsaturated Carbonyl compounds. Woodward-Fieser rules can be extended to calculate the λmax of α,β-unsaturated carbonyl compounds.
Comments are closed.
|
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
